[Authorization to start clinical sessions for a drug for amyotrophic lateral sclerosis]
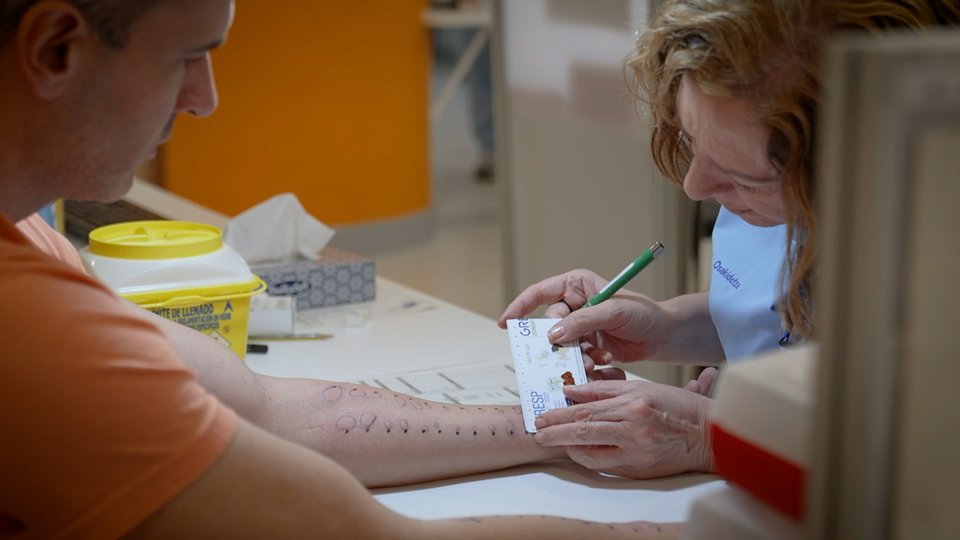
The Spanish Agency for Medicines and Medical Devices (AEMPS) has authorised the testing in people of a potentially effective drug for amyotrophic lateral sclerosis (ALS). The drug is called AP-2 and has been developed at the Margarita Salas Biological Research Center of the CSIC. Testing is expected to begin in April. First, safety and pharmacokinetics will be evaluated in healthy volunteers and the next phase with patients is expected to begin in January 2027.
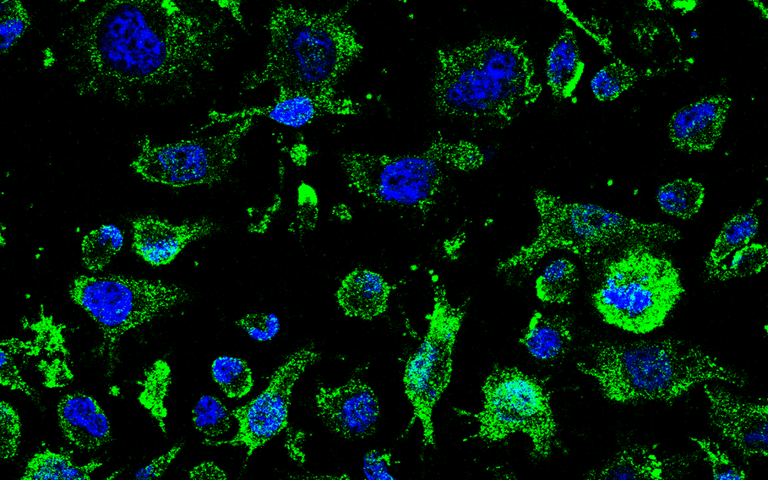
The purpose of the AP-2 drug is to reacquire the function of the TDP-43 protein. The TDP-43 protein is pathologically modified in patients with AEA, causing the death of motor neurons, i.e., cells of the nervous system responsible for transmitting signals from the brain or spinal cord to the muscles. AP-2 has succeeded in reversing the TDP43 abnormality in both cell models and transgenic animals.
“The TDP-43 protein is pathologically modified in patients with AD and causes motor neuron death.”
The researchers say that, although it is still early, reproducing the results obtained in cells and animals could slow down the disease and, at best, stop it.
In Europe, there is only one approved drug for the treatment of MS in Europe: riluzole, a palliative drug that improves symptomatology and prolongs life expectancy by three to six months. Therefore, the authorisation of the AP-2 clinical trial is a promising step forward.
Buletina
Bidali zure helbide elektronikoa eta jaso asteroko buletina zure sarrera-ontzian