An enzyme for cleaning toxins from acrylic materials
To combat the hidden toxicity of acrylic paints, scientists from POLYMAT-UPV have created a clean and effective solution based on a vegetable enzyme.
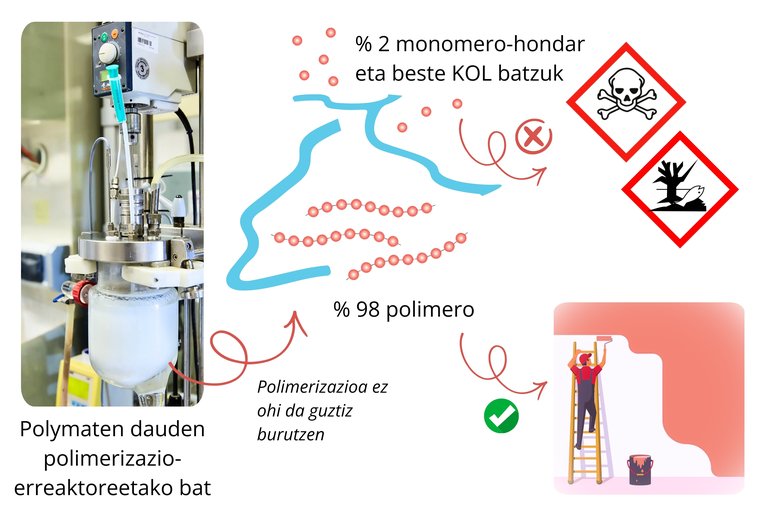
Water-based paints and adhesives are often considered a less toxic alternative to organic solvent-based products, both for health and the environment. The use of water as the main medium significantly reduces polluting emissions and increases safety, both in the manufacturing process and in domestic use.
However, there are still challenges in completely eliminating these water-based (or acrylic) paints in health and the environment. Many people will be familiar with this situation: when painting a room with such a paint, smells appear that can last for days. But if the paint does not contain organic solvents, where do these smells come from?
The answer lies in volatile organic compounds (VOCs). A large amount of these odors come from so-called monomer residues, small molecules that have not completely reacted in the manufacture of paint, adhesive or other polymers1. Although their concentration is very low, of the order of ppm (parts per million), their presence is not innocuous. These levels can also result in unpleasant odors, eye and respiratory irritation, and other adverse health effects during prolonged exposures.
Therefore, European legislation establishes very strict limits for the amounts of these VOCs in paints, coatings and adhesives. With this challenge in mind, researchers at POLYMAT (UPV/EHU) have developed a solution that seeks to improve existing strategies to reduce these compounds: an innovative biotechnological method capable of capturing almost all these unwanted compounds2.
One of the most widely used techniques in the manufacture of water-based paints and adhesives is water-based polymerization. In this process, the monomers are dispersed in water along with an initiator. This initiates a chain reaction whereby the monomers are converted to polymers. The result is a latex, which we usually see as an off-white liquid. However, the reaction is rarely complete, and small amounts of unreacted monomer always remain.
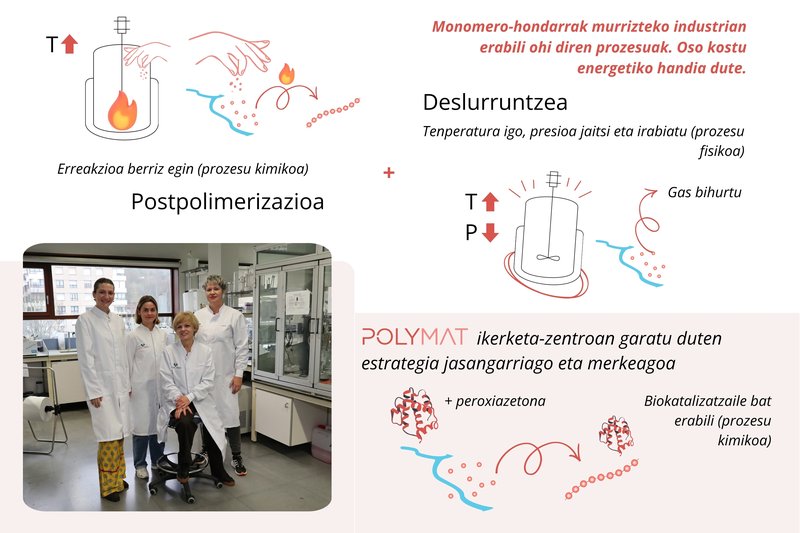
The two main techniques used to reduce monomer residues and the one proposed by Polymat researchers. The following researchers appear in the photo: Ana Trajcheva, Justine Elgoyhen, Oihane Sanz and Radmila Tomovska.
As explained in more detail in another article in the Elhuyar magazine3, at the industrial level there are two strategies to reduce this waste. The first is chemical postpolymerization: after the main reaction is completed, more initiator is added to force the reaction of the remaining monomers. This is a simple method, but since it requires heating, it requires a lot of energy and can produce new unwanted VOCs. The second is devolatilization, in which heat and vacuum are used to pass the monomers and other volatile organic compounds into the gas phase. Although it is very efficient, it has an enormous energy consumption.
The alternative developed in POLYMAT is based on the integration of a biotechnological tool: An enzyme extracted from the roots of Armoracia rusticana (radish). This biomolecule is added to the aqueous mixture after completion of the polymerization reaction and, in the presence of hydrogen peroxide and acetylacetone, initiates a chemical process capable of removing residual monomers, even if present in a very small amount.
In this novel process, the monomer molecules are linked together by a reaction promoted by the enzyme and peroxyacetone. Upon bonding, the monomers form short polymers which are integrated between the polymer chains formed above. The enzyme, on the other hand, the part with the highest value, acts as a catalyst, that is, it is not consumed, which allows to reduce costs. In the experiments carried out, conversions of greater than 95% (percentage of VOC that is removed) have been obtained in the case of methyl methacrylate monomer (MMA) and close to 90% in the case of butyl acrylate (BA). They are the two most common monomers in water-based paints and adhesives.
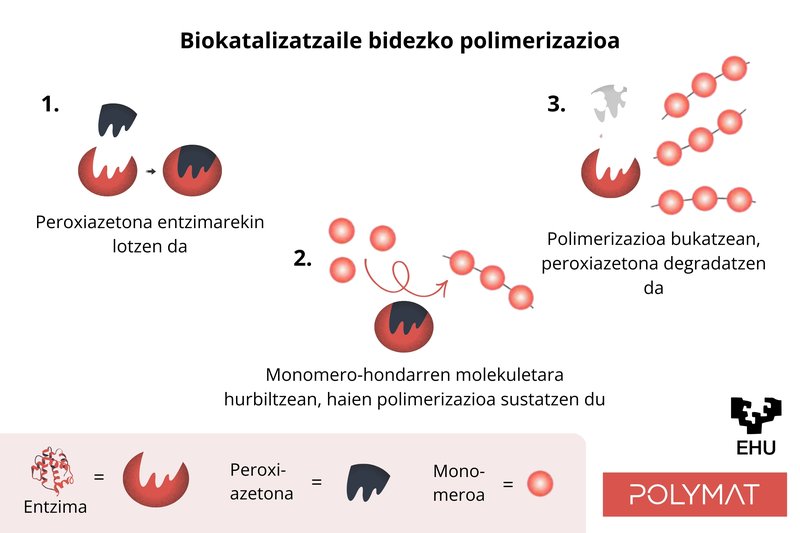
Mechanism by which monomer is converted to polymer using enzyme and peroxyacetone.
A particularly important aspect is that the present method is effective at ambient pressure and ambient temperature and does not produce new VOC-type compounds. This makes it a more sustainable alternative to the other two traditional strategies.
Researchers have found that in addition to reducing VOCs, this process also influences the properties of the polymer. The resulting material has greater flexibility, which may or may not be beneficial depending on the application. This effect is due to the fact that the enzyme and the produced short polymer chains act as plasticizers and facilitate the movement of the polymer chains. On the other hand, an improvement in the thermal stability of the material is observed. Upon heating, the enzyme is carbonized to form a protective layer which delays the degradation of the polymer, which increases its resistance to high temperatures.
Although the results are promising, the process is still in the laboratory phase. The next steps will be aimed at adapting to the industrial scale. If this goal is achieved, an enzyme extracted from a common plant could become an essential tool for the development of more sustainable paints and adhesives.
References
[1] Https://zientzia.eus/artikuluak/zer-dira-polimeroak/
[2] Https://www.sciencedirect.com/science/article/pii/S0014305726000121
[3] Https://zientzia.eus/artikuluak/polimeroen-monomero-hondarrak-murriztu-egin-daitez/
Buletina
Bidali zure helbide elektronikoa eta jaso asteroko buletina zure sarrera-ontzian














